 It is 115.7 degrees Celsius (240.26 degrees Fahrenheit) in temperature. It is due to VSEPR theory that both structures are different. The number of bond pairs and lone pairs combine to give a shape, while the ratio of both determine any varience from the regular geometry. In solid form, the XeF4 has a density of 4.040 g cm3 and has a solid white look. Chemistry Chemistry questions and answers What is the molecular geometry of XeF4 tetrahedral, octahedral, square planar, seesaw, or trigonal bipyramidal This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. In addition, the individual Xe-F bonds are said to be covalent because it shares the electrons, with this the bonds making the molecule nonpolar. Molecular shape of benzene (C6H6) : - bent/v-shaped - linear - octahedral - see-saw - square planar - square pyramidal - tetrahedral - trigonal bipyramidal - trigonal planar - trigonal. Its chemical equation could simply be written as : Xe + 2F2 > XeF4 In this process, elemental fluorine supposedly oxidizes xenon, under some specific conditions of temperature and pressure. This chemical compound is formed when xenon reacts with fluorine. 
Which of the following has a T-shaped structure NH3 BCl3 ICl3 PCl3 SO3 3. The angle between two sp hybrid orbitals. The formal charge for XeF4 is zero, and since the XeF4 molecular geometry is symmetrical due to the bond formation and dipole moments, the molecule is considered nonpolar. XeF4 is the chemical formula of the compound Xenon Tetrafluoride. Which of the following has square-planar molecular geometry SF4 XeF4 CCl2Br2 CCl4 PH3 2. Which series correctly identifies the hybridization of the central atom in a molecule of AlCl3 sp. 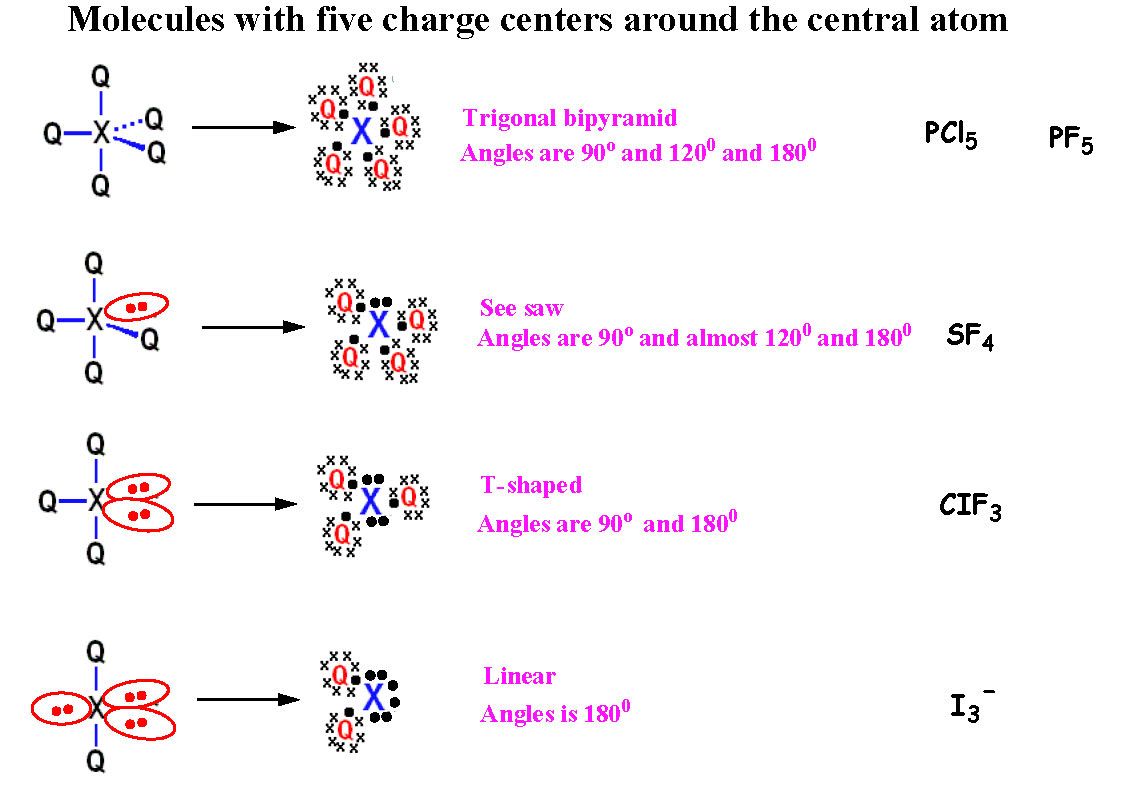
Which of the following has a T-shaped structure NH3. Sb is in group 5, so it has 5 electrons in its outer shell. There are 4 Sb-F bonds, and also a negative charge on the central antinomy atom, so there are in total (5+4+1=) 10 electrons. Therefore there are 5 pairs. This suggests it is a trigonal bipyramedal shape, (seesaw) typically with bond angles of 120 and 90 degrees. However as there are 4 bond pairs, and 1 lone pair, then the lone pair will distort this perfect geometry, forming angles between Sb-F bonds to be less than 90 and 120 degrees. The lone pair forms on the axial part of the molecule as there is minimum repulsion. Which of the following has square-planar molecular geometry SF4. Therefore it has 8 electrons in its outer shell. Fluorine atoms have 7 electrons, yet one is an unpaired electron, so this is the electron in fluorine that gets involved in bonding. There are 4 of these, so around the xenon atom, there are (8+4=) 12 electrons, so 6 electron pairs and as there are 4 fluorine atoms bonding, there are 4 bond pairs, and 2 lone pairs of electrons. Therefore this is an octahedral shape, with 90 degree angles between each bond and lone pair of electrons. Due to VSEPR (valence shell electron pair repulsion) theory, the most energetically stable form of XeF4 will form, with the lone pairs as far away from each other as possible, hence forming the shape seen. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
